Trialynx
AI-powered clinical trial platform that generates ICH and FDA-compliant documents in record time.
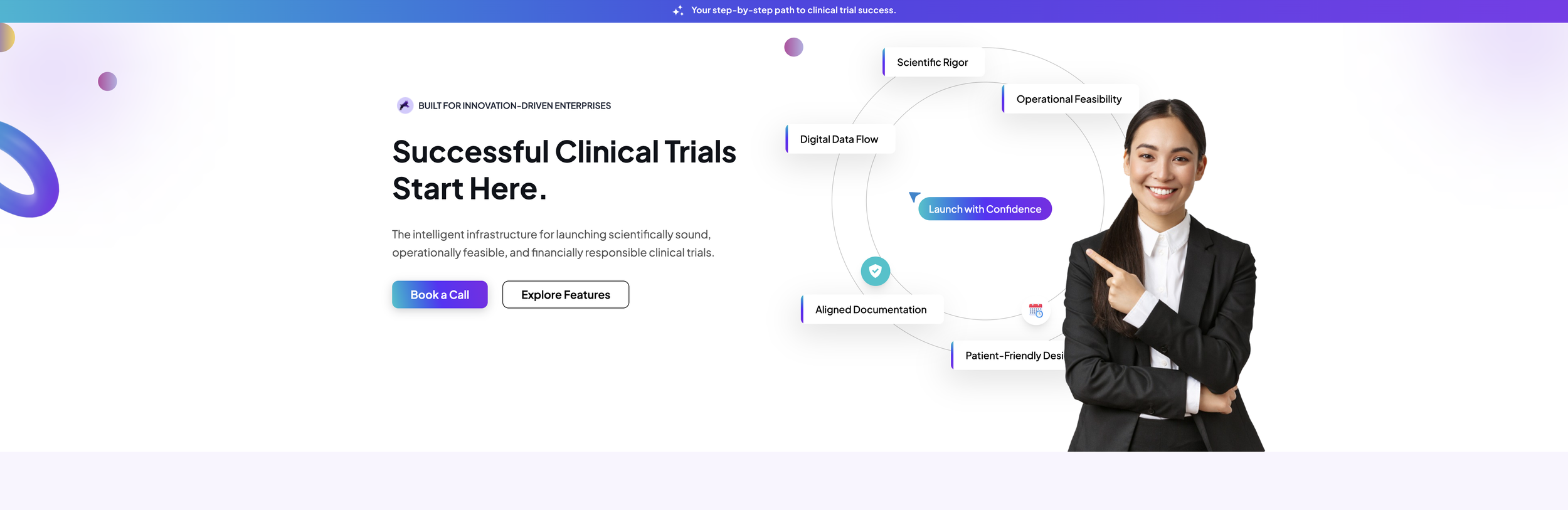
Trialynx has developed a revolutionary digital platform that uses automation and AI to instantly generate protocols, consent forms, and other regulatory documents — allowing researchers to launch clinical trials faster than ever before.
Engage NSS for Trialynx → contact@northstarsci.com
Platform capabilities
Automated ICH and FDA-compliant document generation
AI-driven protocol design and authoring
Informed consent form generation
Regulatory document workflows for sponsors and CROs
Compressed study startup timelines
Who Trialynx is for
Clinical trial sponsors launching new studies
CROs scaling document operations across multiple trials
Clinical operations teams managing protocol amendments
Regulatory affairs teams under timeline pressure
Why North Star Scientific represents Trialynx
NSS is the commercial partner for Trialynx in the United States and Canada. Our team brings 25+ years of life science and clinical trial commercial experience to introducing Trialynx's AI platform to clinical trial sponsors and CROs who need to compress study startup timelines.
What NSS handles for Trialynx:
Outbound prospecting to clinical trial sponsors and CROs
AI-personalized multi-channel outreach across email, LinkedIn, voice, and phone
In-person trade show representation at clinical research conferences
Account-level engagement with pharma sponsors and large CROs
Pipeline management and monthly reporting to the Trialynx commercial team
How to engage
United States and Canada NSS team: Pat Téa, Jeff Storelee, Pearl Orbeta, Nell Bonus Contact: contact@northstarsci.com | 1-888-565-4957
For platform demos, technical specifications, and integration details: